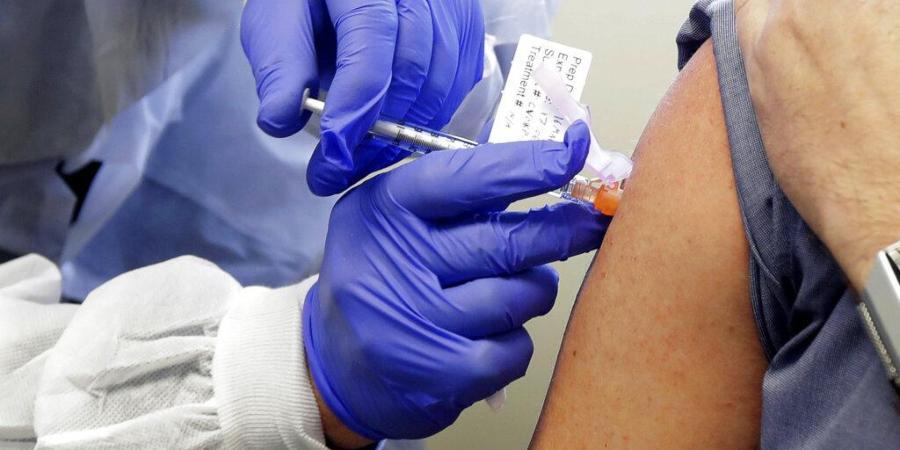
NEW DELHI: At least seven Indian pharma companies are working to develop a vaccine against coronavirus as they join global efforts to find a preventive to check the spread of the deadly virus that has already infected more than 14 million globally.
Bharat Biotech, Serum Institute, Zydus Cadila, Panacea Biotec, Indian Immunologicals, Mynvax and Biological E are among the domestic pharma firms working on the coronavirus vaccines in India.
Vaccines normally require years of testing and additional time to produce at scale, but scientists are hoping to develop a coronavirus vaccine within months because of the pandemic.
Bharat Biotech has received approval to conduct phase I and II clinical trial for its vaccine candidate Covaxin, that has been developed and manufactured in the company’s facility in Hyderabad.
It last week started human clinical trials.
Leading vaccine major Serum Institute of India has said that it is hoping to develop a COVID-19 vaccine by the year-end.
“At present, we are working on the AstraZeneca Oxford vaccine which is undergoing phase III clinical trials. In addition to this, we will also start human trials in India in August 2020. Based on the current situation and most recent updates on the clinical trials, we are hoping that the AstraZeneca Oxford vaccine will be available towards the end of this year,” Serum Institute of India CEO Adar Poonawalla told PTI.
The company is also developing a live attenuated vaccine with US-based biotech firm Codagenix, which is undergoing pre-clinical trials, he added.
“Apart from AstraZeneca Oxford vaccine and Codagenix, we have associated with multiple institutions worldwide as manufacturing partners for vaccine candidates that are being developed. These include Austria’s Themis along with two others,” Poonawalla said.
On the partnership with AstraZeneca, Poonawalla said: “Serum Institute of India has entered a manufacturing partnership with AstraZeneca to produce and supply 1 billion doses of the COVID-19 vaccine being developed by Oxford University.”
These vaccines will be for India and middle and low income countries across the world (GAVI countries), he added.
Pharma major Zydus Cadila has said that it is looking to complete clinical trials of its COVID-19 vaccine candidate ZyCoV-D in seven months.
The company had last week started clinical trials of its COVID-19 vaccine candidate with the first human dosing.
Depending on the study outcomes and if the data is encouraging and the vaccine is found to be effective during the trials, it could take a total of seven months for the trials to be completed and for the vaccine to be launched, Zydus Cadila Chairman Pankaj R Patel said in a statement.
Hyderabad-based Bharat Biotech last week started human trials of its vaccine Covaxin at Rohtak’s Post-Graduate Institute of Medical Sciences.
The phase I and II clinical trials of the vaccine for SARS-CoV-2 by Bharat Biotech have been approved by the Indian drug regulator after pre-clinical studies demonstrated safety and immune response.
The company has developed the vaccine in collaboration with the Indian Council of Medical Research (ICMR) and the National Institute of Virology (NIV).
Panacea Biotec in June said that it was setting up a joint venture firm in Ireland with US-based Refana Inc to develop a vaccine for COVID-19.
The company in partnership with Refana aims to manufacture over 500 million doses of COVID-19 candidate vaccine, with over 40 million doses expected to be available for delivery early next year, Panacea Biotec had said.
Indian Immunologicals, a subsidiary of National Dairy Development Board (NDDB), has inked an agreement with Australia’s Griffith University to develop a vaccine for coronavirus.
Others like Mynvax and Biological E are also working to develop vaccines for COVID-19.
Vaccines typically provide the immune system with harmless copies of an antigen: a portion of the surface of a bacterium or virus that the immune system recognises as foreign.
A vaccine may also provide a non-active version of a toxin – a poison produced by a bacterium – so that the body can devise a defence against it.

They must follow higher safety standards than other drugs because they are given to millions of healthy people.
Vaccine testing is a four-stage process — pre-clinical testing on animals, phase I clinical testing on a small group of people to determine its safety and to learn more about the immune response it provokes, phase II trials are expanded safety trials, and phase III testing is done by administering it to thousands of people to confirm its efficacy.
Globally, the World Health Organisation (WHO) is tracking around 140 candidates vaccines, of which around two dozen are in various phases of human clinical trials.
Chinese company Sinovac Biotech is moving into phase III trials in Brazil while University of Oxford/AstraZeneca is in a combine phase II/III trial in the UK and has recently gone into phase III trials in South Africa and Brazil.
US-based Moderna expects to start phase III trials of its vaccine candidate this month.
Among other leading players, German firm BioNTech is collaborating with pharma major Pfizer to develop a vaccine for COVID-19.
The companies have received fast track designation from the US Food and Drug Administration (USFDA) for two investigational vaccine candidates being developed to help protect against SARS-CoV-2.